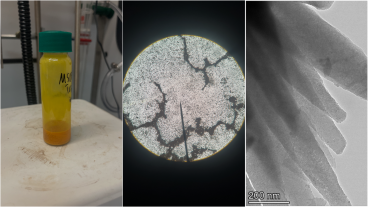
Researchers from the Colorado School of Mines Chemistry Department and the National Renewable Energy Laboratory have developed a perovskite solar cell that retains its efficiency after 1,000 hours of continuous use, with their findings published in Nature Energy.
Associate Professor Alan Sellinger, graduate student Tracy Schloemer and former Mines postdoc Jonathan Tinkham are co-authors of the paper, titled “Tailored interfaces of unencapsulated perovskite solar cells for >1,000 hour operational stability.” The project was led by NREL’s Joseph Luther and Joseph Berry and also included Jeffrey Christians, Philip Schulz, Steven Harvey and Bertrand Tremolet de Villers.
Over the past decade, perovskites have rapidly evolved into a promising technology, now with the ability to convert about 23 percent of sunlight into electricity. But work is still needed to make the devices durable enough for long-term use.
According to the researchers, their new cell was able to generate power even after 1,000 straight hours of testing. While more testing is needed to prove the cells could survive for 20 years or more in the field—the typical lifetime of solar panels—the study represented an important benchmark for determining that perovskite solar cells are more stable than previously thought.
A new molecule developed by Sellinger, nicknamed EH44, was used to replace an organic molecule called spiro-OMeTAD that is typically used in perovskite solar cells. Solar cells that use spiro-OMeTAD experience an almost immediate 20 percent drop in efficiency, which continues to steadily decline as it becomes more unstable.
The researchers theorized that replacing the layer of spiro-OMeTAD could stop the initial drop in efficiency in the cell. The lithium ions within the spiro-OMeTAD film move uncontrollably throughout the device and absorb water. The free movement of the ions and the presence of water causes the cells to degrade. EH44 was incorporated as a replacement because it repels water and doesn’t contain lithium.
The use of EH44 as the top layer resolved the later more gradual degradation but did not solve the initial fast decreases that were seen in the cell’s efficiency. The researchers tried another approach, this time swapping the cell’s bottom layer of titanium dioxide (TiO2) for one with tin oxide (SnO2). With both EH44 and SnO2 in place, as well as stable replacements to the perovskite material and metal electrodes, the solar cell efficiency remained steady. The experiment found that the new SnO2 layer resolved the chemical makeup issues seen in the perovskite layer when deposited onto the original TiO2 film.
“This study reveals how to make the devices far more stable,” Luther said. “It shows us that each of the layers in the cell can play an important role in degradation, not just the active perovskite layer.”
Funding for the research came from the U.S. Department of Energy Solar Energy Technologies Office.
CONTACT
Mark Ramirez, Managing Editor, Communications and Marketing | 303-273-3088 | ramirez@mines.edu
Emilie Rusch, Public Information Specialist, Communications and Marketing | 303-273-3361 | erusch@mines.edu