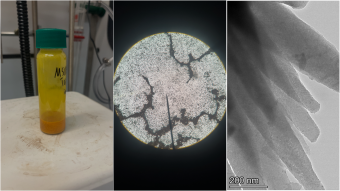
A sample of Trip3Tez-I shown in a vial, viewed with an optical microscope under 10x magnification and viewed using a transmission electron microscope (TEM).
An international team of researchers led by Colorado School of Mines has demonstrated a new way to assemble molecules into porous materials.
Chalcogen bonding, a recently recognized category of noncovalent bonding, has been applied in catalysis and molecular assembly studies, but its impact on material behavior and properties remains poorly understood.
In findings published today in the journal Nature Synthesis, researchers showed for the first time that chalcogen bonding can be used to build permanently porous materials conceptually akin to metal-organic frameworks (MOFs), which won the Nobel Prize in Chemistry in 2025. The team also found that these honeycomb-like molecular materials have some unexpected properties, particularly their potential use as semiconductors.
“This is just the tip of the iceberg,” said corresponding author C. Michael McGuirk, associate professor of chemistry at Colorado School of Mines. “We think this is the first discovery that's going to potentially precipitate a whole new field of materials chemistry."
To the naked eye, the permanently porous chalcogen-bonded organic framework (ChOF) created by the researchers – called Trip3Tez-I – looks like an orange powder. But when examined with electron diffraction, researchers were able to confirm not only the existence of two nanometer-wide permanent pores in the “molecular sponge” but also that the singular interaction holding the material together is chalcogen bonds between tellurium and nitrogen atoms, said coauthor Hannah Martin, a PhD candidate in materials science at Mines.
“This gives us a really interesting platform to understand how this bonding interaction, chalcogen bonding, impacts material properties,” Martin said.
According to computations, those potential properties include the ability to conduct electricity in a controllable manner. Acting as a semiconductor isn’t unique in and of itself – there are many MOFs that are also semiconductors – but Martin said what is interesting are the additional capabilities introduced by the weaker chalcogen bonds.
“With many other materials that have semiconducting properties, once you make it, it’s stuck and often the only way to dissolve the bonds is basically melting it in acid,” she said. “One of the main reasons that people are really interested in noncovalent interactions like chalcogen bonding and hydrogen bonding because the interactions are a lot weaker – you can regrow your materials, you can regrow your crystals, redissolve them, reform them."
In the future, those regenerative properties could open the door to healable molecular semiconductors with better charge transport and more, McGuirk said.
“Instead of building semiconductors from the top down through a fabrication process, you could design molecules and then spontaneously assemble them into these semiconductors in a much more modular approach,” he said.
Also contributing to the international effort were researchers from University of Oregon, North Carolina State University, University of Southampton (UK), National Institute of Standards and Technology, National Laboratory of the Rockies, Oak Ridge National Laboratory and Rigaku Americas.
Read the full study, “A Permanently Porous Chalcogen-Bonded Organic Framework,” at https://www.nature.com/articles/s44160-026-01072-x/.




