Samaniuk wins NSF CAREER Award for work on 2D particle interaction, fabrication

Joseph Samaniuk, assistant professor in the Department of Chemical and Biological Engineering at Colorado School of Mines, has received a National Science Foundation CAREER Award for work to improve the understanding of interactions between ultra-thin, 2D particles.
Atomically thin particles – described that way because they are typically only one to three atoms thick – are of interest to scientists because of the unique properties that such small thickness creates. Electrical conductivity, mechanical strength and optical transparency can all be significantly enhanced in these 2D materials compared with their bulk material counterparts.
Samaniuk will receive $534,043 over five years for his project, “Interactions between 2D particles at fluid-fluid interfaces,” which looks specifically at how 2D particles interact with each other when confined to a fluid-fluid interface and seeks to develop techniques for fabricating model 2D particles of controlled size and shape.
Here, Samaniuk answers a few questions about this fundamental research and how it could potentially impact the future of photovoltaics.
Q: What is your latest research focused on?
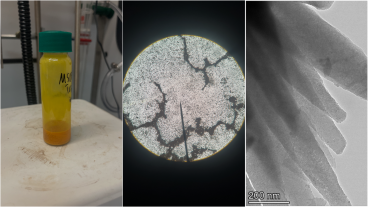
A: We’re looking at particles that are atomically thin, one or two or three atoms thick but also very long and very wide, almost like a sheet of paper. We call them 2D particles because of this, and we’re looking at how these 2D particles interact when you put them down at a fluid-fluid interface. Think about a bowl of milk and Cheerios – the Cheerios will stick together side to side. Think of that, but the Cheerios are 2D particles. Understanding why and how they interact in that way is important if you want to create an atomically thick material by assembling many of these particles side to side.
Graphene is one of the most common examples of a 2D particle – it is very, very conductive along the length of the particle, but it’s also very transparent because of how thin it is. If you put a bunch of graphene particles down at a fluid-fluid interface, you can’t see them, but when the particles are assembled side by side, they form a very conductive film.
Q: What do you find most exciting about your research?
This really gets at very fundamental work in colloids and interfaces. In my lab we do a lot of applied work, and also a bit of fundamental work. This is a great opportunity to dive into very fundamental physics. To me, that’s very exciting. I like work that is very applied as well, where you could do some work and it would be instantly relevant to industry. This particular project isn’t like that – it’s relevant at a fundamental scientific level.
Q: What is the potential impact of this work?
We see this project and understanding this system as a potential route to making large quantities of low-cost films for photovoltaics, or more generally what are called optoelectronic devices—devices that take optics and integrate them with electronics. It’s essentially what a photovoltaic cell does. It has the potential ability to lower the cost of photovoltaics and integrate with other electronics.
In this project, we've proposed to look at a number of 2D particles, including graphene, hexagonal boron nitride (h-BN) and molybdenum disulfide (MoS2). Hexagonal boron nitride has a similar atomic structure as graphene, although with boron and nitrogen instead of carbon, but it’s not electrically conductive. You can combine materials like graphene and hexagonal boron nitride to make layers of conductive and nonconductive material. It’s what you do in traditional electronic devices but this is at the atomic level.
You could populate pages and pages and pages of 2D particles that people have synthesized. The reason many people are excited about 2D materials is you can make them with so many different properties. Because they are atomically thick but very, very large laterally, they have really unique properties compared to bulk materials. Graphene is just a sheet of carbon. Graphite is its bulk analogue. But because it’s atomically thin, graphene has unique properties.
Q: How does this research agenda inform your teaching?
An integral part of this grant is working with SUMMET, the Summer Multicultural Engineering Training Program at Mines. Built into this grant is the proposal to work with SUMMET over the next couple of years by bringing in demonstrations to try to convey the science underlying the work in this proposal, and by informing the high school students who participate in this program about future opportunities in undergraduate research. We also plan to track how much the students take away from the demos and discussion. We want to quantify how much talking about the science and learning about undergraduate research may influence the students’ choices to stay in STEM to pursue graduate degrees. We’re trying to take surveys of the students early on, and there’s an effort to follow up with the students, year after year. That will be a challenging effort, but we’ll give it a go to see how much this particular intervention will stick with them.